How Carbon Filters Work for Drinking Water.
Carbon granuals water filtering is a method to remove contaminants and impurities, removing chlorine, sediment, volatile organic compounds (VOCs), taste and odor from water.
Carbon granuals water filtering is a method to remove contaminants and impurities, removing chlorine, sediment, volatile organic compounds (VOCs), taste and odor from water.
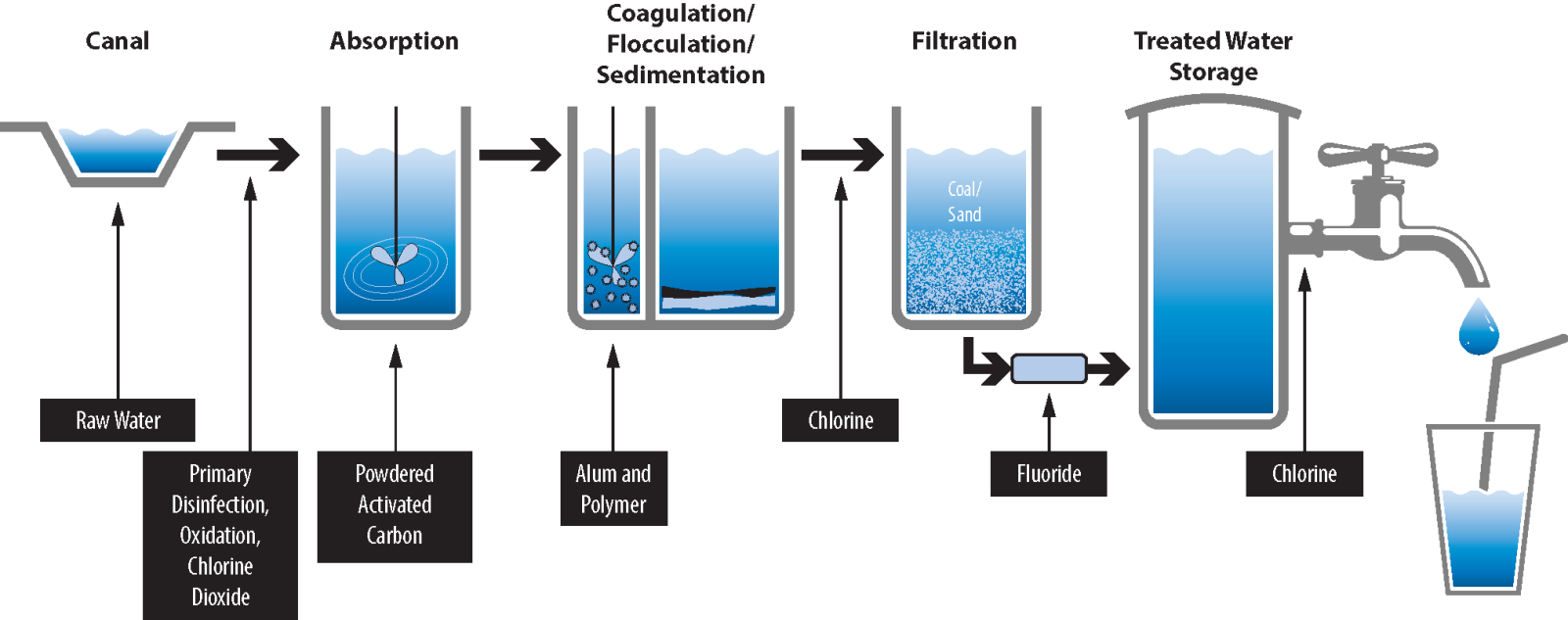
Activated carbon is commonly used to adsorb natural organic compounds, taste and odor compounds, and synthetic organic chemicals in drinking water treatment. Adsorption is both the physical and chemical process of accumulating a substance at the interface between liquid and solids phases. Activated carbon is an effective adsorbent because it is a highly porous material and provides a large surface area to which contaminants may adsorb. The two main types of activated carbon used in water treatment applications are granular activated carbon (GAC) and powdered activated carbon (PAC).
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations Na+and hydroxide anions OH−Sodium hydroxide is a highly caustic base and alkali that decomposes proteins at ordinary ambient temperatures and may cause severe chemical burns. It is highly soluble in water and readily absorbs moisture and carbon dioxide from the air. It forms a series of hydrates NaOH·nH2O. The monohydrate NaOH·H2O crystallizes from water solutions between 12.3 and 61.8 °C.